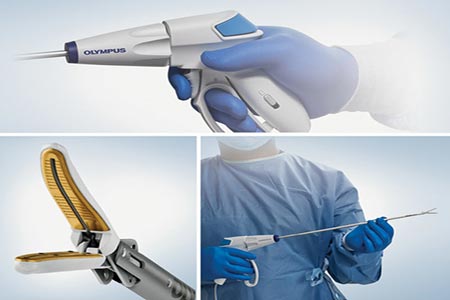
Olympus was the first launch of the devices in the new POWERSEAL™ family of advanced bipolar surgical energy products.
The POWERSEAL 5mm Curved Jaw Tissue Sealer and Divider, Double-Action devices meet the highest standards of clinical performance for advanced bipolar surgical energy devices by delivering consistent sealing reliability in an ergonomic, multifunctional design that promotes procedural efficiency.
With the release of the POWERSEAL devices, Olympus is introducing a line of versatile advanced bipolar surgical energy devices that augment an extensive and differentiated surgical energy portfolio, which features the unique THUNDERBEAT™ hybrid energy devices and SONICBEAT™ ultrasonic dissectors.
The POWERSEAL devices position Olympus competitively within the growing global market for advanced bipolar surgical energy devices, currently estimated at over $1.2B USD. [i]
The POWERSEAL devices provide surgeons with state-of-the-art sealing, dissection, and grasping capabilities in laparoscopic or open surgery while greatly reducing the force required by the surgeon to close the jaws.
[ii] The POWERSEAL devices can be used in numerous forms of surgical intervention including general surgery and gynecological, colorectal, bariatric, urological, thoracic, and vascular surgical procedures.
"Initial clinical experience has shown that the new 5mm POWERSEAL devices offer uniform tissue compression for consistently strong sealing," said Justin A. Maykel, MD, Chief of Colon and Rectal Surgery at UMass Memorial Health. "The product design makes the devices both intuitive and safe to use."
"We are thrilled to expand our surgical energy product portfolio with best-in-class advanced bipolar energy solutions that were designed with surgeons' needs and clinical outcomes as the primary drivers," said Phil Roy, Global Vice President and General Manager for Surgical Devices in the Therapeutic Solutions Division of Olympus Corporation.
"These POWERSEAL devices were developed with a core requirement of zero compromise in meeting our goals of strength, efficiency and physician comfort. Testing showed we exceeded our goals across the key measures of surgical energy device performance such as sealing speed, reliability and thermal profile, all while delivering a superior user experience." [v]
As outlined in the Instructions for Use, reusing or re-sterilizing the POWERSEAL devices can cause excessive wear and degradation of device performance as well as possible cross-contamination, which may potentially affect patient safety. Use of the POWERSEAL devices with non-Olympus generators may result in undesired tissue effect or injury to the patient or to the surgical team.
The POWERSEAL devices should not be used in patients who have electronic implants without first consulting a cardiologist. Other risks are associated with lack of training, use with an energized endoscope that is not a Type CF applied part, use with a patient with vascular pathology and use at high current for long durations.
The Olympus Energy platform, made up of the ESG-400™ and USG-400™ electrosurgical generators, offers hospitals a complete energy solution for any operating room and powers the full range of Olympus Energy systems: monopolar, bipolar (HiQ ™, HICURA ™, PK ™ and Everest ™ devices), ultrasonic (SONICBEAT devices), hybrid energy (THUNDERBEAT devices) and advanced bipolar (POWERSEAL devices).
By operating from one energy platform, hospitals can reduce capital expenditures while meeting the other goals of the Quadruple aim[vi], availing themselves of a range of surgical energy technologies that have been designed with both the patient and the physician in mind.
Advanced bipolar energy devices are distinguished from traditional bipolar energy devices through more efficient energy delivery, intuitive tissue monitoring, and uniform pressure applied to the tissue by the device jaws. These attributes enable Advanced Bipolar technologies to deliver more consistent and larger (7mm) vessel sealing than standard bipolar technology counterparts.[iii]
The POWERSEAL devices are regulatory approved for sale in the U.S., Puerto Rico and Australia at this time, and are actively under review in numerous other global markets.
For more information, visit medical.olympusamerica.com.