Are we prescribing antibiotics wrong?

Outsourcing Clinical Trials Outside of the US

Regulators And Standards Groups Take Steps To Address Emerging Technologies In Biopharma

Two-dimensional liquid chromatography with multiple heart-cutting
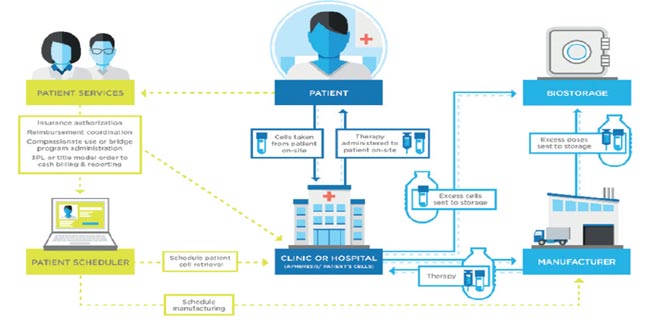
Understanding Pharmaceutical Logistics Validation in a Dynamic Environment





Latest White Papers
- Random
Featured News
Latest News
- Title
- Most Views
- Recently Added
- Ordering
- Random



Virax Biolabs Group Limited an innovative biotechnology company focused on immunology research and ...

Wegovy® HD (semaglutide) injection 7.2 mg is available now nationwide, delivering the highest ...

Acute liver injury (ALI), often triggered by conditions such as acetaminophen overdose, can rapidly ...

BD a leading global medical technology company, and Ypsomed, a leading developer of injection ...



Latest Updates
Kivu Bioscience, a clinical-stage biotechnology company developing next-generation antibody-drug conjugates (ADCs) for difficult-to-treat cancers, announced the appointment of Mohit Trikha, Ph.D., as ...
Kivu Bioscience, a clinical-stage biotechnology company developing next-generation antibody-drug conjugates (ADCs) for difficult-to-treat cancers, announced the appointment of Mohit Trikha, Ph.D., as Chief Executive Officer. Dr. Trikha, who previously served as President and Chief Operating ...
OrthoFX™, a leader in innovative clear aligner technology, announced preliminary results from a retrospective clinical study comparing treatment effectiveness and predictability of its FXClear ...
OrthoFX™, a leader in innovative clear aligner technology, announced preliminary results from a retrospective clinical study comparing treatment effectiveness and predictability of its FXClear aligner system with the leading clear aligner brand, Invisalign. The study, titled "Treatment ...
Dexcel Pharma USA is pleased to announce that it has received final approval from the U.S. Food and Drug Administration (FDA) for its Abbreviated New Drug Application (ANDA) for Nintedanib Capsules, ...
Dexcel Pharma USA is pleased to announce that it has received final approval from the U.S. Food and Drug Administration (FDA) for its Abbreviated New Drug Application (ANDA) for Nintedanib Capsules, 100 mg and 150 mg, the generic equivalent of OFEV® (nintedanib) capsules1. Nintedanib is indicated ...
Johnson & Johnson a worldwide leader in multiple myeloma therapies, announced that the U.S. Food and Drug Administration (FDA) approved TECVAYLI® plus DARZALEX FASPRO® (daratumumab and ...
Johnson & Johnson a worldwide leader in multiple myeloma therapies, announced that the U.S. Food and Drug Administration (FDA) approved TECVAYLI® plus DARZALEX FASPRO® (daratumumab and hyaluronidase-fihj) for the treatment of adults with relapsed or refractory multiple myeloma (RRMM) who have ...
BD a leading global medical technology company, announced it has obtained CE Marking for the Revello™ Vascular Covered Stent, a next-generation endovascular solution for the treatment of ...
BD a leading global medical technology company, announced it has obtained CE Marking for the Revello™ Vascular Covered Stent, a next-generation endovascular solution for the treatment of atherosclerotic lesions in the common and external iliac arteries.
This advancement arrives at a pivotal ...
Parnell announced the launch of Pentobarbital Sodium and Phenytoin Sodium Euthanasia Solution, a Class III controlled veterinary product formulated for humane, painless, and rapid euthanasia of dogs, ...
Parnell announced the launch of Pentobarbital Sodium and Phenytoin Sodium Euthanasia Solution, a Class III controlled veterinary product formulated for humane, painless, and rapid euthanasia of dogs, available in 100 mL multi‑dose vials.
Designed for clinical reliability and ease of ...