The Naval Medical Research Center (NMRC) and The Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc.
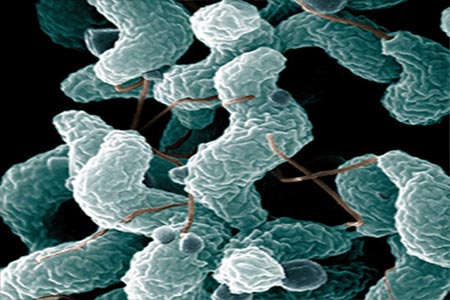
(HJF) recently established an agreement with Lumen Bioscience, located in Seattle, to develop a new immunotherapy to prevent gastrointestinal diseases caused by Campylobacter jejuni and enterotoxigenic E. coli (ETEC).
Both pathogens can cause diarrhea and dysentery in deployed military service members and other travelers, including children.
Diarrhea kills 2,195 children every day—more than AIDS, malaria, and measles combined, according to the US Centers for Disease Control and Prevention (CDC). The occurrence of these diseases is most prevalent in areas with limited water treatment facilities. Although the military has developed extensive capabilities for the provision of clean food and water, diarrhea has continued to be a problem for deployed personnel.
The goal of this project is to develop an ultra-low cost, orally delivered biologic to prevent infection. The biologic would be composed of a blue-green algae spirulina product that incorporates Campylobacter- and ETEC-specific tiny variable domains on a heavy chain antibodies (VHHs) to prevent diarrhea. A VHH is derived from a special type of antibody naturally produced by camelids, a group of animals that includes alpacas, camels, and llamas. Their small size allows engineering of spirulina to express the VHHs to develop a product that provides pathogen-specific protection through antibody expression.
The development and testing of this new Lumen product is being funded by Combating Antibiotic Resistant Bacteria Biopharmaceutical Accelerator (CARB-X). CARB-X is a global nonprofit partnership led by Boston University and dedicated to funding the development of new products to address antibiotic-resistant bacteria. Lumen has received an initial funding commitment of $5.2M, with the project eligible for an additional $8.3M if project milestones are met, subject to availability of funds from CARB-X. Of this full amount, Lumen will pay NMRC/HJF up to $2.5M to support testing of the effectiveness of the Lumen spirulina product. NMRC will test the product's effectiveness combating antibiotics resistance by testing directly against the pathogen strains through the development of animal models of disease with the eventual goal of testing effectiveness in humans.
"This initiative is a truly important collaborative effort to develop an anti-diarrheal product for the warfighter and children in countries where these diarrheal diseases are endemic. By capitalizing on our expertise in developing preventive therapies against Campylobacter and ETEC, and with Lumen's innovative spirulina technology, we will be able to quickly progress this anti-diarrheal product from bench to bedside." said Dr. Renee Laird, an HJF employee working in support of NMRC.
Utilizing bioinformatics, NMRC/HJF will identify Campylobacter and ETEC strains to be used in the manufacture of the spirulina product. Scientists will identify samples of 200 C. jejuni and 200 ETEC isolates and test for the prevalence of resistance against the most common antibiotics used for the treatment of Infectious Diarrhea. NMRC/HJF will also develop and use a mouse infection challenge model to test Lumen's products' ability to prevent disease. The final year of the award will continue with animal testing and the preparation of regulatory filings to determine if the spirulina product is effective in humans.